This is "Unit 4", section 4.3 from the book General Chemistry (v. 1.0).
4.3 Names and Formulas of Binary Molecular Compounds
Learning Objective
- To name molecular compounds with two elements.
So far in this unit, our focus has been on the bonds and interactions between the atoms. In this section and the next we turn our attention to nomenclature. These substances we know to be collections of atoms have names. From the names of the compounds, we know their formulas. This section introduces us to the nomenclature of molecular compounds. Section 4.4 introduces the nomenclature of ionic compounds.
Before going any further it is critical that you know the difference between molecular and ionic compounds. Review Section 4.2. Compounds between two nonmetals are molecular. Compounds between a metal and a nonmetal are ionic.
Binary Molecular Compounds
Binary molecular compounds—that is, covalent compounds that contain only two nonmetal elements—are named with a system utilizing Greek prefixes to indicate the number of atoms of each kind. The procedure, diagrammed in Figure 4.3(1), uses the following steps:
Figure 4.3(1) Naming a Covalent Inorganic Compound

Place the elements in their proper order.
- The element farthest to the left in the periodic table is usually named first. If both elements are in the same group, the element closer to the bottom of the column is named first.
- The second element is named with the the suffix -ide attached to the root of the element name.
Identify the number of each type of atom present.
Prefixes derived from Greek stems are used to indicate the number of each type of atom in the formula unit (Table 4.3(1) ). The prefix mono- (“one”) is used only when absolutely necessary to avoid confusion, just as we omit the subscript 1 when writing molecular formulas.
To demonstrate steps 1 and 2a, we name HCl as hydrogen chloride (because hydrogen is to the left of chlorine in the periodic table) and PCl5 as phosphorus pentachloride. The order of the elements in the name of BrF3, bromine trifluoride, is determined by the fact that bromine lies below fluorine in group 17.
Table 4.3(1) Prefixes for Indicating the Number of Atoms in Chemical Names
Prefix Number mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10 undeca- 11 dodeca- 12 - If a molecule contains more than one atom of both elements, then prefixes are used for both. Thus N2O3 is dinitrogen trioxide, as shown in Figure 4.3(1).
- In some names, the final a or o of the prefix is dropped to avoid awkward pronunciation. Thus OsO4 is osmium tetroxide rather than osmium tetraoxide.
***Certain compounds are always called by the
common names that were assigned long ago when names rather than
formulas were used. For example, H2O is
water (not dihydrogen monoxide); NH3 is
ammonia.
Note the Pattern
Start with the element at the far left in the periodic table and work to the right. If two or more elements are in the same group, start with the bottom element and work up.
Example 4.3-1
Write the name of each binary covalent compound.
- SF6
- N2O4
- ClO2
Given: molecular formula
Asked for: name of compound
Strategy:
List the elements in order according to their positions in the periodic table. Identify the number of each type of atom in the chemical formula and then use Table 4.3(1) to determine the prefixes needed.
Solution:
- Because sulfur is to the left of fluorine in the periodic table, sulfur is named first. Because there is only one sulfur atom in the formula, no prefix is needed. There are, however, six fluorine atoms, so we use the prefix for six: hexa- (Table 4.3(1) ). The compound is sulfur hexafluoride.
- Because nitrogen is to the left of oxygen in the periodic table, nitrogen is named first. Because more than one atom of each element is present, prefixes are needed to indicate the number of atoms of each. According to Table 4.3(1) , the prefix for two is di-, and the prefix for four is tetra-. The compound is dinitrogen tetroxide (omitting the a in tetra- according to step 2c) and is used as a component of some rocket fuels.
- Chlorine is named first because ClO2 is an oxide of an element other than fluorine. A prefix is not necessary because each molecule has only one atom of chlorine. Because there are two oxygen atoms, the compound is a dioxide. Thus the compound is chlorine dioxide. It is widely used as a substitute for chlorine in municipal water treatment plants because, unlike chlorine, it does not react with organic compounds in water to produce potentially toxic chlorinated compounds.
Exercise
Write the name of each binary covalent compound.
- IF7
- N2O5
- OF2
Answer:
- iodine heptafluoride
- dinitrogen pentoxide
- oxygen difluoride
Example 4.3-2
Write the formula for each binary covalent compound.
- sulfur trioxide
- diiodine pentoxide
Given: name of compound
Asked for: formula
Strategy:
List the elements in the same order as in the formula, use Table 4.3(1) to identify the number of each type of atom present, and then indicate this quantity as a subscript to the right of that element when writing the formula.
Solution:
- Sulfur has no prefix, which means that each molecule has only one sulfur atom. The prefix tri- indicates that there are three oxygen atoms. The formula is therefore SO3. Sulfur trioxide is produced industrially in huge amounts as an intermediate in the synthesis of sulfuric acid.
- The prefix di- tells you that each molecule has two iodine atoms, and the prefix penta- indicates that there are five oxygen atoms. The formula is thus I2O5, a compound used to remove carbon monoxide from air in respirators.
Exercise
Write the formula for each binary covalent compound.
- silicon tetrachloride
- disulfur decafluoride
Answer:
- SiCl4
- S2F10
The structures of some of the compounds in Example 4.3-1 and Example 4.3-2 are shown in Figure 4.3(2) "The Structures of Some Covalent Inorganic Compounds and the Locations of the “Central Atoms” in the Periodic Table", along with the location of the “central atom” of each compound in the periodic table. It may seem that the compositions and structures of such compounds are entirely random, but this is not true.
Figure 4.3(2) The Structures of Some Covalent Inorganic Compounds and the Locations of the “Central Atoms” in the Periodic Table
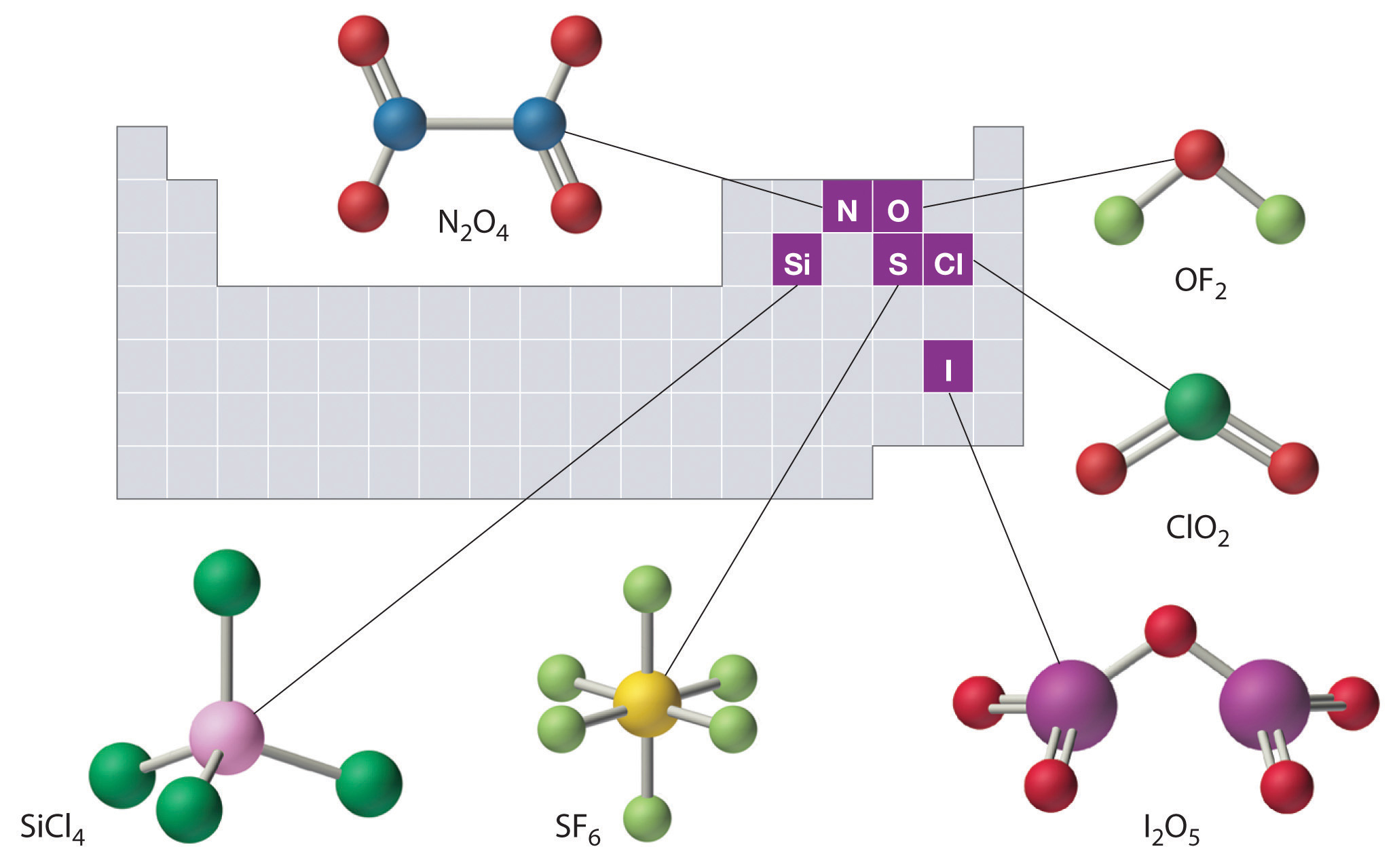
The compositions and structures of covalent inorganic compounds are not random. As you will learn in future chapters, they can be predicted from the locations of the component atoms in the periodic table.
Summary
Covalent inorganic compounds are named using prefixes to indicate the numbers of atoms in the molecular formula.
Conceptual Problems
-
Name each compound.
- NO
- NO2
- N2O
- CS2
- SO3
- NF3
- SF6
-
Name each compound.
- NCl3
- IF5
- N2O5
- Cl2O
- P2S5
- PCl5
-
Would you expect PCl3 to be an ionic compound or a covalent compound? Explain your reasoning.
Answer
-
- nitrogen monoxide
- nitrogen dioxide
- dinitrogen monoxide
- carbon disulfide
- sulfur trioxide
- nitrogen trifluoride
- sulfur hexafluoride
-
-
Numerical Problems
-
Write the formula for each compound.
- dinitrogen monoxide
- silicon tetrafluoride
- boron trichloride
- nitrogen trifluoride
- phosphorus tribromide
-
Write the formula for each compound.
- dinitrogen trioxide
- iodine pentafluoride
- boron tribromide
- oxygen difluoride
- arsenic trichloride
Answers
-
- N2O
- SiF4
- BCl3
- NF3
- PBr3
-





